Chapter 1 · Introduction
1.3 Properties
Properties are measurable characteristics used to describe the state of a thermodynamic system. Common properties include pressure, temperature, volume, and mass. These properties are generally classified as either extensive or intensive.
An extensive property depends on the amount of matter or the size of the system. Examples include total mass and total volume. An intensive property is independent of the amount of matter in the system. Intensive properties remain unchanged when a system is divided into smaller parts, whereas extensive properties scale with system size.
In this section, we focus on three important properties in thermodynamics: specific volume, temperature, and pressure.

1.3.1 Specific Volume
Specific volume is defined as the volume occupied by a unit mass of a substance. It is an intensive property because it does not depend on the total amount of material—only on how much volume each unit of mass occupies.
This diagram illustrates the concept of specific volume for a gas (air) in two identical containers, each with a total volume of 2 m³. In the first container, 1 kg of air occupies the volume, resulting in a specific volume of 2 m³/kg. In the second container, 4 kg of air occupies the same volume, resulting in a lower specific volume of 0.5 m³/kg. Because gases are compressible, more mass can fit into the same space, which leads to higher density and lower specific volume as more mass is added.

In thermodynamics, we frequently work with specific properties, which are values expressed per unit mass. By convention, specific properties are denoted with lowercase letters. For example, total volume is represented as V, while specific volume is represented as v. Other specific properties—such as specific internal energy (u) and specific enthalpy (h)—will be introduced as needed.
Specific volume is calculated from the total volume and mass using
Since density (\(\rho\)) is defined as mass per unit volume, specific volume is simply the inverse of density:
1.3.2 Temperature
Temperature is an intensive property that indicates how “hot” or “cold” a substance is.
Thermal Equilibrium
When two bodies at different temperatures are placed in contact, heat flows from the hotter body to the colder one. This continues until both reach the same temperature. At that point, no more heat flows between them. The bodies are in thermal equilibrium.
Zeroth Law of Thermodynamics
The Zeroth Law formalizes thermal equilibrium and allows us to define and measure temperature using thermometers. If two objects are both in equilibrium with the same thermometer, we know they are also in equilibrium with each other—even if they never touch.
If two bodies are each in thermal equilibrium with a third body, they are in thermal equilibrium with each other. For example, if two bodies have the same temperature as measured by a thermometer, then they have the same temperature as each other.
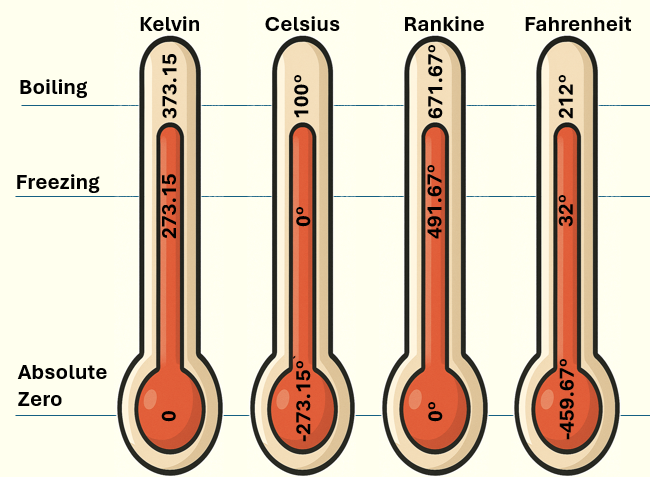
Temperature Scales
In everyday life, we commonly use degrees Celsius (°C) and degrees Fahrenheit (°F). However, in thermodynamics, absolute temperature scales are often used, where zero represents absolute zero—the lowest possible temperature:
- Kelvin (K) — absolute zero is −273.15 °C
- Rankine (°R) — absolute zero is −459.67 °F
Comparison of temperature scales showing absolute zero, freezing point, and boiling point of water under standard atmospheric pressure (1 atm). Each thermometer shows the equivalent values for Kelvin, Celsius, Rankine, and Fahrenheit.
Because many problems in thermodynamics require temperature to be expressed on an absolute scale, it's common to convert from Celsius or Fahrenheit to their respective absolute counterparts using the following formulas:
1.3.3 Pressure
Pressure is defined as force per area:
Where:
- P is pressure
- F is the force applied perpendicular to a surface
- A is the area over which the force is applied
Pressure is a scalar quantity and is measured in pascals (Pa) in the SI system, where:
Common Pressure Units
Since the pascal is a small unit for most practical applications, larger or more convenient units are commonly used:
- Kilopascal (kPa): 1 kPa = 1,000 Pa
- Megapascal (MPa): 1 MPa = 1,000,000 Pa
- Pounds per square inch (psi): used in the U.S. Customary system
- Bar: 1 bar = 100,000 Pa
- Atmosphere (atm): 1 atm = 101,325 Pa
Types of Pressure
Understanding pressure can be confusing because it depends on the reference point used. The three commonly encountered types are:
- Absolute Pressure: Pressure measured relative to absolute zero pressure (a complete vacuum). It includes atmospheric pressure.
- Gauge Pressure: Pressure measured relative to atmospheric pressure. This is the reading most pressure gauges display.
- Vacuum Pressure: Pressure below atmospheric pressure, often described as negative gauge pressure.
Relationship between absolute pressure (\(P_{\text{abs}}\)), atmospheric pressure (\(P_{\text{atm}}\)), gauge pressure (\(P_{\text{gauge}}\)), and vacuum pressure (\(P_{\text{vac}}\)). Absolute pressure is measured relative to a perfect vacuum (\(P_{\text{abs}} = 0\)), while gauge pressure and vacuum pressure are measured relative to atmospheric pressure.