Chapter 1 · Introduction
1.4 Systems
A system is a defined region in space selected for analysis. Everything outside of the system is referred to as the surroundings. The surface that separates the system from its surroundings is called the boundary, which can be fixed (stationary) or movable.
In thermodynamics, how we choose the system and its boundary determines how we apply mass and energy balances. Two common categories are closed systems (control mass) and open systems (control volumes).
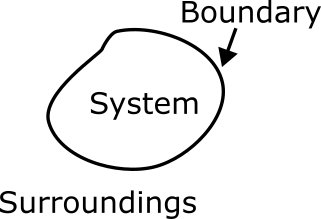
1.4.1 What Is a System?
A system is the specific region or quantity of matter selected for analysis. Everything outside this region is the surroundings. The boundary separates the system from the surroundings and may be real or imaginary, fixed or movable.
Diagram illustrating the components of a thermodynamic system. The system is the region under study, enclosed by a boundary that separates it from the surroundings. The boundary may be real or imaginary, and either fixed or movable, depending on the context of the problem.

In practice, system boundaries are chosen to simplify modeling while capturing the essential physics. A good system definition makes the conservation of mass and energy easier to apply.
1.4.2 Closed Systems (Control Mass)
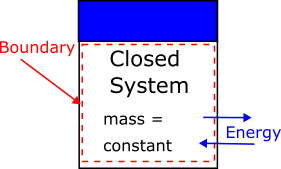
A closed system, or control mass, contains a fixed amount of mass. No mass crosses the system boundary, but energy in the form of heat or work may enter or leave. The volume of a closed system may change, such as in a piston–cylinder device.
Example of a closed thermodynamic system. The system boundary (shown as a dashed line) encloses a fixed amount of mass. Energy in the form of heat or work can cross the boundary, but mass cannot.
If neither mass nor energy crosses the boundary, the system is called an isolated system. Isolated systems are idealizations used to model situations where interactions with the surroundings are negligible.
1.4.3 Open Systems (Control Volumes)
An open system, or control volume, allows both mass and energy to cross the boundary. Open systems are used in problems involving flowing fluids, such as:
- Nozzles and diffusers
- Turbines
- Compressors and pumps
- Heat exchangers
The boundary of an open system is often called the control surface. It may be fixed or movable and can be real (a physical surface, such as a pipe wall) or imaginary (a conceptual surface drawn to define where mass and energy cross).
Illustration of real and imaginary boundaries in an open thermodynamic system. Real boundaries correspond to physical surfaces, such as the wall of a pipe or a container. Imaginary boundaries are conceptual control surfaces used to define regions where mass and energy cross the system boundary.
